2010
| News | Registration | Abstracts | Accommodation | Excursions | Deadlines | Organizing committee |
| First circular | Participants | Abstract submission | Travel | Program | Seminar History | Contact us |
| Новости |
| Первый циркуляр |
| Регистрация |
| Оформление тезисов |
| Тезисы |
| Программа |
| Участники |
| Размещение |
| Экскурсии |
| Проезд |
| Важные даты |
| Оргкомитет |
| Обратная связь |
Ba-Ti-rich phogopite from olivine melaleucitites of the Urmia Lake region, Iran
Sharygin V.V.*, Kryvdik S.G.**
* V.S.Sobolev Institute of Geology and Mineralogy SB RAS, Novosibirsk, Russia; ** N.P.Semenenko Institute of Geochemistry, Mineralogy and Ore Formation NAS of Ukraine, Kyiv, Ukraine, kryvdik@ukr.net
The first data about the presence and chemistry of specific Ba-Ti mica in olivine melaleucitites from the Neogene volcano on the north-east cost of the Urmia Lake, Iran were previously documented by Krivdik and Mikhailov (2007). In addition to olivine melaleucitites this volcano is also comprised by leucitites, leucite phonolites, tephrites and their tuffs and trachytes (Krivdik, Mikhailov, 2007). Olivine melaleucitites contain abundant phenocrysts (up to 40 vol.%) of clinopyroxene (SiO2 - 47.2-53.5; TiO2 -0.1-1.9; Al2O3 - 1.1-5.1; FeOt - 4.0-7.9; MgO - 15.2-17.1; Na2O - 0.2-0.7 wt.%) and leucite-pseudoleucite, phenocrystal olivine (Fo82-84, CaO - 0.4-0.5 wt.%) is minor. The groundmass consists of clinopyroxene, leucite, Ti-magnetite (TiO2 - 8.5-9.3 wt.%), Ba-Ti-mica, fluorapatite and K-feldspar, nepheline and sodalite occur rarely. It should be noted that groundmass micas enriched in both Ba and Ti were previously described in olivine nephelinites and related rocks around the world (Mansker et al., 1979; Edgar, 1992; Zhang et al., 1993; Seifert, Kampf, 1994; Shaw, Penczak, 1996; Greenwood, 1998; Kogarko et al., 2005; Sharygin, 2009). Here we present new data on chemistry and substitutions of Ba-Ti-mica from the Urmia olivine melaleucitites.
Brown Ba-Ti-mica forms xenomorphic grains in the groundmass and occurs as a phase of polycrystalline intergrowths or a daughter phase of melt inclusions in phenocrystal clinopyroxene and leucite. The inclusions together with individual crystallites of leucite, olivine, nepheline and apatite commonly decorate the growth zones in the host clinopyroxene. Ba-Ti-mica from inclusions is associated with silicate glass, Ti-magnetite, apatite, sodalite and zeolite-like minerals. According to EMPA data most micas contain high BaO (8.2-13.9 wt.%), TiO2 (4.4-9.4 wt.%) and F (2.7-5.1 wt.%) (Table 1). Only rare grains in clinopyroxene show low content of these components. In general, most mica grains do not show significant variations in chemical composition. The core-to-rim variations in some grains indicate the slight enrichment in K2O and depletion in BaO.
Table 1. Chemical composition (EMPA, wt.%) of Ba-Ti-phlogopites from the Urmia olivine melaleucitites.
|
Sample |
I-5 |
|
|
|
|
|
I-8 |
|
|
|
|
|
|
|
|
Position |
gm |
|
gm |
|
in Cpx |
in Cpx |
gm |
|
in Plc |
|
in Plc |
in Cpx |
|
in Cpx |
|
|
c |
r |
c |
r |
|
|
c |
r |
c |
r |
|
c |
r |
|
|
SiO2 |
32.02 |
33.73 |
30.77 |
32.72 |
31.48 |
34.37 |
29.88 |
30.79 |
29.49 |
29.15 |
36.38 |
31.80 |
31.42 |
37.41 |
|
TiO2 |
6.04 |
5.91 |
6.23 |
5.67 |
6.37 |
5.51 |
6.33 |
5.43 |
6.90 |
6.94 |
4.59 |
5.10 |
4.57 |
2.84 |
|
Nb2O5 |
0.00 |
0.00 |
0.00 |
0.01 |
0.00 |
0.06 |
0.01 |
0.02 |
0.00 |
0.02 |
0.02 |
0.00 |
0.04 |
0.00 |
|
Cr2O3 |
0.02 |
0.01 |
0.00 |
0.01 |
0.05 |
0.04 |
0.02 |
0.02 |
0.00 |
0.00 |
0.02 |
0.06 |
0.05 |
0.45 |
|
Al2O3 |
13.67 |
13.00 |
14.21 |
13.76 |
14.34 |
13.03 |
15.32 |
15.33 |
15.39 |
15.37 |
12.33 |
15.08 |
14.75 |
14.56 |
|
Fe2O3* |
4.36 |
4.19 |
3.95 |
3.98 |
3.50 |
3.09 |
3.35 |
2.63 |
3.11 |
3.30 |
0.85 |
1.39 |
2.91 |
0.50 |
|
FeO* |
4.86 |
4.83 |
4.89 |
4.68 |
6.48 |
6.84 |
6.54 |
5.85 |
8.19 |
8.40 |
10.87 |
8.01 |
6.85 |
8.52 |
|
MnO |
0.12 |
0.13 |
0.12 |
0.14 |
0.19 |
0.13 |
0.18 |
0.11 |
0.20 |
0.27 |
0.34 |
0.13 |
0.16 |
0.09 |
|
MgO |
19.16 |
19.49 |
18.37 |
19.75 |
17.55 |
17.89 |
17.32 |
18.54 |
15.58 |
15.29 |
15.01 |
16.66 |
18.56 |
21.10 |
|
CaO |
0.04 |
0.07 |
0.03 |
0.02 |
0.09 |
0.26 |
0.02 |
0.03 |
0.04 |
0.03 |
0.18 |
0.27 |
0.28 |
0.07 |
|
BaO |
9.81 |
8.12 |
11.31 |
9.29 |
10.34 |
7.80 |
12.25 |
11.69 |
12.03 |
12.24 |
7.16 |
12.55 |
11.69 |
0.81 |
|
Na2O |
0.70 |
0.71 |
0.63 |
0.72 |
0.70 |
0.91 |
0.57 |
0.59 |
0.61 |
0.62 |
0.85 |
0.62 |
0.63 |
0.37 |
|
K2O |
5.50 |
6.16 |
5.01 |
5.79 |
4.94 |
6.18 |
4.75 |
4.91 |
4.59 |
4.59 |
6.35 |
4.26 |
4.91 |
9.38 |
|
F |
4.73 |
4.49 |
4.50 |
4.57 |
3.73 |
5.06 |
3.81 |
4.27 |
3.75 |
3.98 |
5.19 |
3.97 |
4.06 |
0.30 |
|
Cl |
0.01 |
0.00 |
0.00 |
0.01 |
0.02 |
0.00 |
0.01 |
0.01 |
0.00 |
0.00 |
0.00 |
0.03 |
0.04 |
0.03 |
|
H2O* |
1.12 |
1.31 |
1.20 |
1.27 |
1.66 |
1.15 |
1.60 |
1.49 |
1.62 |
1.47 |
1.31 |
1.75 |
1.56 |
3.91 |
|
Total |
102.16 |
102.15 |
101.23 |
102.39 |
101.43 |
102.32 |
101.95 |
101.70 |
101.49 |
101.67 |
101.44 |
101.66 |
102.48 |
100.33 |
|
O=(F,Cl)2 |
1.99 |
1.89 |
1.90 |
1.93 |
1.57 |
2.13 |
1.61 |
1.80 |
1.58 |
1.68 |
2.19 |
1.68 |
1.72 |
0.13 |
|
Total |
100.17 |
100.26 |
99.33 |
100.46 |
99.85 |
100.19 |
100.34 |
99.90 |
99.91 |
100.00 |
99.26 |
99.99 |
100.76 |
100.19 |
Note: gm - groundmass; c, r - core and rim of grain; in Cpx - silicate melt and polycrystalline inclusions in clinopyroxene phenocrysts; in Plc - silicate melt and polycrystalline inclusions in leucite or pseudoleucite. * - calculated from formula on the basis of 11 oxygens, Fe2O3 is estimated only as tetrahedral Fe3+ in formula.
The formulas calculated on the basis of 11 oxygens indicate that composition of the Urmia micas varies from phlogopite K(Mg,Fe)3[AlSi3O10](OH)2 to barian titanian phlogopite and fluorphlogopite (K,Ba)(Mg,Fe,Ti)3 [AlSi3O10](OH,F)2. Most compositions are close to formula (K0.6-0.8Ba0.2-0.4)(Mg1.5-2.3Fe2+0.3-1.0Ti0.2-0.5)[Fe3+0.1-0.3Al1.1-1.4Si2.3-2.7O10](F0.8-1.3OH0.7-1.2). In general, compositions of the Iran micas are different from oxykinoshitalite, other Ba-Ti-rich oxymicas and micas from alkaline rocks (Mansker et al., 1979; Zhang et al., 1993; Kogarko et al., 2005; Sharygin, 2009) in higher Ba, H2O and F and lower Ti. The positive correlation of Ba with Al and antipathetic relations of Ba with K and Si are shown that K+Si « Ba+Al is a main substitution, whereas isomorphic schemes with participation of Ti are minor (Fig. 1).
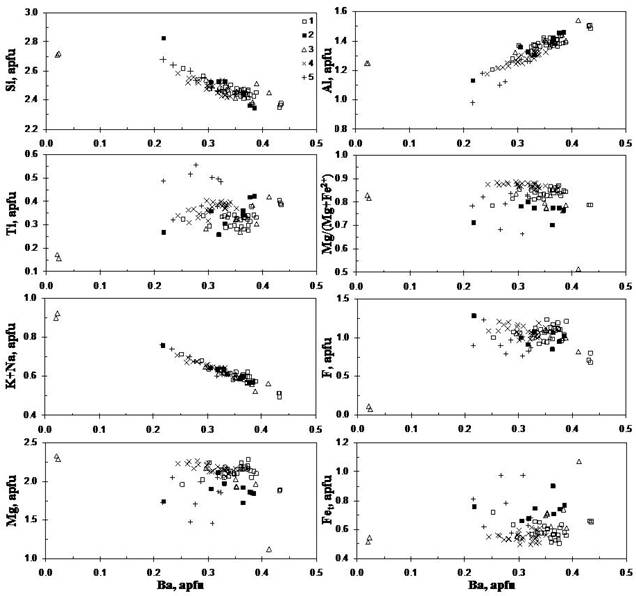
Figure 1. Compositional variations (in apfu) for Ba-Ti-micas from the Urmia olivine melaleucitites.
Note: 1-3 - sample I-8: 1 - groundmass; 2 - inclusions in leucite/pseudoleucite; 3 - inclusions in clinopyroxene; 4-5 - sample I-5; 4 - groundmass; 5 - inclusions in clinopyroxene. Formula is calculated on the basis of 11 oxygens.
The calculated values of H2O in groundmass Ba-Ti-micas of the Urmia Lake melaleucitites strongly suggest that during crystallization initial magma was getting richer in H2O. Such phenomenon probably provoked to transform leucite into pleudoleucite aggregate.
This study was financially supported by RFBR (grant 08-05-00134).
References:
Edgar A.D. Barium-rich phlogopite and biotite from some Quaternary alkali mafic lavas, West Eifel, Germany // European Journal of Mineralogy. 1992. Vol. 4. P. 321-330.
Greenwood J.C. Barian-titanian micas from Ilha da Trindade, South Atlantic // Mineralogical Magazine. 1998. Vol. 62. P. 687-695.
Kogarko L.I., Uvarova Yu.A., Sokolova E., Hawthorne F.C., Ottolini L., Grice J.D. Oxykinoshitalite, a new species of mica from Fernando de Noronha Island, Pernambuco, Brazil: occurrence and crystal structure // Canadian Mineralogist. 2005. Vol. 43. P. 1501-1510.
Krivdik S.G., Mikhailov V.A. Geochemical and mineralogical features of potassic rocks of the Urmia Lake, Iran // In: Shcherbak N.P. (Ed.), Alkaline magmatism of the Earth and its ore potential. Kiev, Logos. 2007. P. 133-134 (in Russian).
Mansker W.L., Ewing R.C., Keil K. Barian-titanian biotites in nephelinites from Oahu, Hawaii // American Mineralogist. 1979. Vol. 64. P. 156-159.
Seifert W., Kampf H. Ba-enrichment in phlogopite of a nephelinite from Bohemia // European Journal of Mineralogy. 1994. Vol. 6. P. 497-502.
Sharygin V.V. Ba-Ti-oxymicas from olivine melanephelinites of the Udokan lava field, Siberia, Russia: chemistry and substitutions // In: Abstracts of XXVI International conference, School “Geochemistry of alkaline rocks”. Moscow, ONTI GEOKHI RAS. 2009. P. 132-134.
Shaw C.S.J., Penczak R.S. Barium- and titanium-rich biotite and phlogopite from the Western and Eastern Gabbro, Coldwell alkaline complex, northwestern Ontario // Canadian Mineralogist. 1996. Vol. 34. P. 967-975.
Zhang M., Suddaby P., Thompson R.N., Dungan M. Barian titanian phlogopite from potassic lavas in northeast China: Chemistry, substitutions, and paragenesis // American Mineralogist. 1993. Vol. 78. P. 1056-1065.