2010
| News | Registration | Abstracts | Accommodation | Excursions | Deadlines | Organizing committee |
| First circular | Participants | Abstract submission | Travel | Program | Seminar History | Contact us |
| Новости |
| Первый циркуляр |
| Регистрация |
| Оформление тезисов |
| Тезисы |
| Программа |
| Участники |
| Размещение |
| Экскурсии |
| Проезд |
| Важные даты |
| Оргкомитет |
| Обратная связь |
GEOCHEMICAL ZONING OF ORE BODY
OF KIYA-SHALTYR DEPOSIT (Kyznetskiy Alatay)
Sazonov Anatoliy, Vulf Marina, Leont’ev Sergey
Siberian Federal University, Krasnoyarsk
According to geological and geochemical features found hidden primary crystallization banding of urtite body, sulphide lentiform and veins in urtites, due to the origin differentiation and liquation of magma at the silicate and sulfide components. Conclusions on elements hidden primary crystallization banding found expression in digital form, confirmed by the results of geochemical modeling, determine the number and measure, which opens up new opportunities for scientific analysis and prediction.
Study of geochemical zoning Kiya-Shaltyr deposit holds the data on the chemical composition of ores. The database includes about 100,000 well samples (horizons from I to XX) and characterizes the urtites ore body from the surface to 150 - 200 m depth, the serial samples in the benching band 5 - 7 m or more, between some - 10 meters.
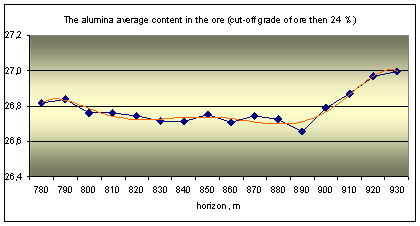
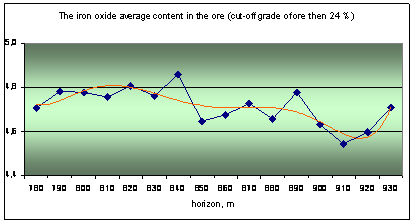
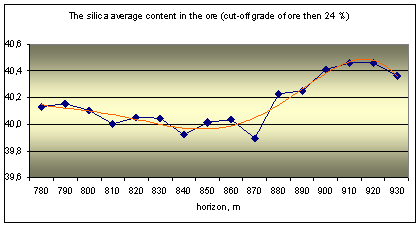
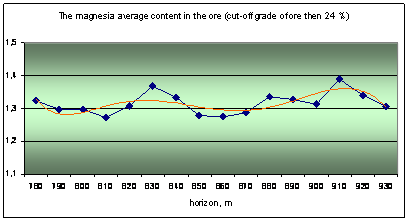
The actual material is data about quality of nepheline ore, obtained during mining deposit. The ore quality is determined by the silicate analysis, it content 9 components: Al2O3, Na2O, K2O, SiO2, Fe2O3, CaO, SO3, MgO and ppp (loss on calcination). The main ore component is alumina and its content are three ore grade: high-alumina (28%), common (28 - 26,5%) and low-alumina (26,5 - 24%).
The original database is processed by mathematical statistics methods, the results are shown in the graphs as the distribution to depth of main ore components (Fig. 1).
|
|
|
|
|
|
|
Fig. 1 Distribution of ore components in depth |
|
Analysis of the graphs shows the rhythmic distribution of elements and confirms the primary crystallization banding is there. It was distinguished by a way multidimensional regression simulation modeling methods and wavelet transformation of geochemical data about urtite body.
The cycles of different level and scale from 20-30 to 50-60 and 120-130 meters can be distinguished on the graphs. Some ore components have similar trends and they are united into two groups as results of correlation analysis: 1) Al2O3, Na2O, K2O and SiO2; 2) Fe2O3, CaO, SO3, MgO and ppp. Elements of the first group (aluminous) are part of nepheline, it’s the main ore minerals associated with rich ores. Elements of the second group (ferruginous) are part of titanavgite, sulfides and carbonates are associated with poor ores.
The complexity index is
calculated and graphs are drawn for these groups of elements (Fig.
2). The complexity index is calculated by the formula:
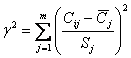
where
![]() -
is the
complexity index,
-
is the
complexity index,
![]() -
the content of the j-th element in the i-th point,
-
the content of the j-th element in the i-th point,
![]() -
the average
arithmetic contents of the j-th element,
-
the average
arithmetic contents of the j-th element,
![]() -
the standard
deviation of the j-th element, m - the number of complexed elements.
-
the standard
deviation of the j-th element, m - the number of complexed elements.
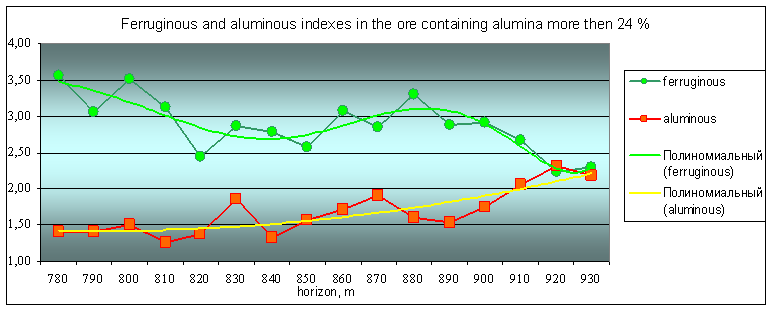
Fig. 2 Alumina and ferric complex indicators
Geochemical deposit model shows the frequency distribution of major components of ores in a vertical section. The general trend is to change the rhythmic peaks of total concentrations of ferruginous association (Fe2O3, CaO, SO3, MgO) and aluminous associations (Al2O3, Na2O, K2O, SiO2). In general, the concentration of alumina components decreases to depth of the ore body (from 930 to 780 of the horizon), and iron - is increasing. The rhythms boundaries we can make allocate at 790-800, 850-860 and 920-930 m.
Geological significance of alumina and iron associations are rhythmic changes deals with the processes of differentiation. Differentiation evidence manifested repeatedly at all levels of the geological structure this object.
First, differentiation in the deep magma reservoir, which was formed as a result the alkali and alkaline basaltic magma intrusion Kuznetskiy Alatay.
Secondly, differentiation in magmatic camera formed Kiya-Shaltyrsk massif, several intrusions consist a single magma series: mezokratic gabbro - leucocratic gabbro - urtite - ijolites dykes and nepheline syenite - veins and lenses-shaped of sulphides.
Thirdly, the urtites body has zonal structure as the depth such along strike and across the strike. This rhythm is shown earlier in the graphs (see Fig. 1 and 2).
Fourth, the differentiation commonly seen in minerals: agpaitic texture of urtites, teardrop-shaped inclusions in sulfides urtites and the structure disintegration of solid solution in the ore minerals (magnetite - ilmenite, pyrrhotite - pentlandite).
The high content of volatiles in the magma (sulfur, hydrocarbons, carbon dioxide, fluorine, chlorine and water) contributed to a more complete course of processes of differentiation and the formation of structure agpaitic urtites. It’s calculated the average agpaitic coefficient for benching band of the ore body is equal to 0.78, which gradually increases with depth.
The magnitude of the agpaitic coefficient in Kiya-Shaltyr urtite are miaskitic nepheline syenites (Na2O+ K2O/Al2O3≤1). In these rocks, pyroxenes should be more idiomorphic then nepheline. In this case, this rule is violated: nepheline is more idiomorphic then pyroxene, i.e. crystallization order of pathogenic minerals consistent agpaitic rocks in which the ratio is greater than unity agpaitic (Na2O + K2O/Al2O3> 1). This is possible only at elevated (up to 3%) of volatile components, which are defined in the ore as loss on calcinations (PPP). Viscosity is very important characteristic of silicate melts which change dramatically by the influence of volatile components.
The urtites of Kiya-Shaltyrskoe deposits contain from 1 - 3 to 10% volatile components, which can serve as catalysts for the differentiation processes and to allow the crystallization of magmas agpaitic by increasing the interval of crystallization, lowering the viscosity and better flow differentiation.
In addition, the sulphides teardrop-shaped inclusions, mainly pyrrhotite, in the amount of 1-3% observed in the whole urtites body volume deeper horizon 920 - 930 m, and sometimes form lenses and veins of massive sulfides and massive texture.
Thus, the differentiation processes and liquation was clearly manifested in the formation of Kiya-Shaltyr deposit and the ore body urtites. The main catalysts for magmatic melt liquid segregation were fluids, which were carrying a large amount of readily soluble in these metals.