2010
| News | Registration | Abstracts | Accommodation | Excursions | Deadlines | Organizing committee |
| First circular | Participants | Abstract submission | Travel | Program | Seminar History | Contact us |
| Новости |
| Первый циркуляр |
| Регистрация |
| Оформление тезисов |
| Тезисы |
| Программа |
| Участники |
| Размещение |
| Экскурсии |
| Проезд |
| Важные даты |
| Оргкомитет |
| Обратная связь |
Abnormal europium behavior at formation of aqueous-chloride fluids during uprising and degassing of granite magmas
Lukanin O.A., Dernov-Pegarev V.F.
Vernadsky Institute of Geochemistry and Analytical Chemistry RAS, Moscow, Russia
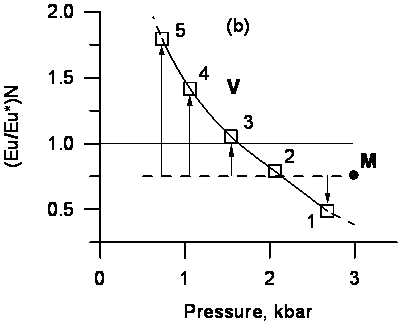
One of the important features of REE behavior in the process of decompressional degassing of granite melts is the presence of europium anomalies in REE spectrum of exsolving fluid phase. Negative Eu anomaly may take place in REE spectrum of fluids enriched by chlorine that were formed under high pressures at early stages of degassing relative to REE spectrum of granite melts. Negative Eu anomaly in fluid is replaced by positive one with pressure decrease and decline of Cl concentration in fluid (Fig.1) (Reed et al.2000; Lukanin, Dernov-Pegarev, 2007, 2010).
|
|
|
|
Fig. 1. Concentration and spectrum REE change in aqueous-chloride fluid formed in the process of decompressional degassing of granite melt at the pressure decrease from 3 to 0.7 kbar. The results of numerical modelling of magmatic degassing in the closed conditions (without removal of a fluid from system) for granite melt having initial contents 7.6 wt.% Н2О and 0.25 wt. % Cl, and also typical for granites concentrations of REE (ppm): 43 La, 83 Ce, 44 Nd, 8.5 Sm, 2 Eu, 7.4 Gd, 1.06 Tb, 1.27 But, 3.25 Yb, 3.25 Lu. a - REE spectra in a fluid at different stages of degassing (Т~800оС) on the various depths corresponding pressures (kbar): 1 – 2.68; 2 2.06; 3 1.54; 4 – 1.06; 5 - 0.73. A dash line (M) – REE spectrum in initial melt. REE concentrations in a melt and a fluid normalized on REE concentrations in chondrites. b - Change of europium anomaly (Eu/Eu*)N in a fluid phase during degassing. A solid line – fluid phase, a dash line – melt (M). Arrows show a difference of (Eu/Eu*)N values in the melt and in the fluid at different stages (depths) of degassing. |
|
Thus, granite melts in the process of degassing during their ascent on different depths can be a source of magmatic fluids with the various concentrations and spectra of REE essentially differing from REE spectra of melt phase. Interaction of these fluids with adjacent rocks can be reflected in REE spectrum of metasomatically changed and vein rocks formed near to granitoid intrusions and subvolcanic bodies.
Observable unique features of europium redistribution between fluid and melt find an explanation in such a fact that Eu in contrast to the other REE under oxidation-reduction conditions, being typical for magmatic process, is present in acidic silica-alumina melts in two valent forms ‑ Eu3 + and Eu2 + ‑ whereas the dominant form for the other REE in such a melts is (REE)3+ (Drake,1975; Wilke, Behrens, 1999).
From the analysis of melt-fluid exchange reactions with participation of two valent forms of europium Eu3 + and Eu2 + follows that the total distribution coefficient of Eu between fluid and melt D(Eu)f/m is equal as a first approximation to (Lukanin, 2008; Lukanin, Dernov-Pegarev, 2010):
D(Eu)f/m = а1 α [C(Cl)f]3 + a2 (1 - α) [C(Cl)f]2,
where C(Cl)f – the concentration of Cl in fluid, α = Eu3+/(Eu3+ + Eu2+), i.e. fraction of Eu3+ in the general amount of europium in the melt, and, a1 and a2 – constants that can be approximately calculated from empirical data upon Eu fluid/melt distribution (Reed et al., 2000).
The equation allows to estimate the influence of oxidation states of europium on sign and value of Eu anomaly, which is expressed by Eu/Eu# ratio, where Eu is real concentration of europium in fluid being in equilibrium with melt with constant Eu3+/(Eu3+ + Eu2+) ratio, and Eu# is possible "virtual" concentration of europium that could be in the same fluid provided that all europium as other REE as well were exclusively present in trivalent form. The sign and value of Eu anomaly in fluid depends upon Cl concentration in fluid and Eu3+/Eu2+ ratio in melt (Fig.2).
|
|
|
Fig. 2. Influence of Eu oxidation states in the melt on value of europium anomaly in fluids with various chlorine concentrations (Lukanin, Dernov-Pegarev, 2010). |
Numbers – Eu3+/(Eu3++ Eu2+), a dash line – a calculated trajectory by results of numerical modeling on the basis of experimental data.
The abnormal behavior of Eu shows itself the stronger, the lower fO2 and, accordingly, the more fraction of Eu2+ is present in melt. It is necessary to notice, that in natural granitoid melts the α value can vary over a wide range, approximately from 0.5 up to 0.8-0.9 (an average ~ 0.7). It corresponds to variations fO2 from NNO-2 to NNO+3 at temperatures which are defined for these melts.
This study was financially supported by RFBR grant 08-05-00022-a and Geosciences Department of the Russian Academy of Science (the program 2- 2010).
References:
Reed M. J., Candela P.A., Piccoli P.M. The distribution of rare earth elements between monzogranitic melt and the aqueous volatile phase in experimental investigations at 800 C and 200 MPa // Contr. Miner. Petrol. 2000 . V.140. P. 251-261.
Lukanin О.А., Dernov-Pegarev V.F. Redistribution of europium and others rare earth elements between melt and water chlorine-bearing fluid during decompression degassing of granite magmas. //Vestnik Otd. Nauk o Zemle RAN. No 1(25)'2007 URL: http://www.scgis.ru/russian/cp1251/h_dgggms/1-2007/informbul-1_2007/term-30e.pdf
Drake M.J. The oxidation state of europium as an indicator of oxygen fugacity // Geochim. Cosmochim. Acta. 1975. V. 39. P. 55-64.
Wilke M. Behrens H. The dependence of the partitioning of iron and europium between plagioclase and hydrous tonalitic melts on oxygen fugacity // Contrib. Mineral. Petrol. 1999. V. 137. P. 102-114.
Lukanin О.А. The reason of abnormal behavior of europium during degassing of granitic magmas. // Vestnik Otd. Nauk o Zemle RAN, No 1(26)'2008. URL: http://www.scgis.ru/russian/cp1251/h_dgggms/1-2008/informbul-1_2008/magm-20e.pdf
Lukanin О.А., Dernov-Pegarev V.F. Distribution rare-earth elements between aqueous-chloride fluids and melt in the process of granite magmas degassing caused by pressure decrease // Geochemistry International, No 10, 2010.