2010
| News | Registration | Abstracts | Accommodation | Excursions | Deadlines | Organizing committee |
| First circular | Participants | Abstract submission | Travel | Program | Seminar History | Contact us |
| Новости |
| Первый циркуляр |
| Регистрация |
| Оформление тезисов |
| Тезисы |
| Программа |
| Участники |
| Размещение |
| Экскурсии |
| Проезд |
| Важные даты |
| Оргкомитет |
| Обратная связь |
Differentiation of tantalum and niobium in the fluid-magmatic (alkaline and granitic) systems (experimental study)
Chevychelov V.Yu.
Institute of Experimental Mineralogy, Chernogolovka, Moscow oblast, Russia
chev@iem.ac.ru
Many questions of principle of genesis of rare-metal deposits connected with granitoid and alkaline magmatism till now remain insufficiently clear. Experimental researches can help to receive more precise representations about physical and chemical conditions of their formation (Zaraisky, 2004; Chevychelov et al., 2005). Geological data testify to the important role of magmatic factors at formation of albitite, greisen and other genetic types of rare-metal deposits of Ta, Nb, Sn, W, Mo, etc. Owing to their chemical properties these metals have high oxygen affinity and affinity to magmatic melt therefore they collect in residual melt, enriching with themselves late differentiates of evolutionary granite series. Such deposits as a rule are connected with highly differentiated multiphase granite plutons. For modelling of possible conditions of formation of tantalum and niobium deposits of similar type in the present report obtained quantitative data on solubility of columbite (Mn,Fe)(Nb,Ta)2O6 in magmatic melts of various alkalinity and alumina-rich and on partitioning of Ta and Nb during fluid-magmatic interaction are considered.
It is established that the melt composition has the very strong effect on columbite solubility. Its solubility is maximal in alkaline melt (up to ~2 wt % Ta and up to ~5 wt % Nb at P =100 MPa). In the subaluminous melt, the maximum Ta and Nb contents decreased for the order both more and at last the contents of these elements may be in 2-6 times lower in the alumina-rich melt. The alkaline melt showed higher Nb content, whereas the peraluminous melt is relatively enriched in Ta (fig. 1). The temperature dependence of solubility is positive but less pronounced than the effect of melt composition. It is most significant in the subaluminous melt, in which the maximum Ta and Nb contents decreased from ~0.5-0.8 to 0.14-0.05 wt %, respectively, as the temperature decreased from 850 to 650оС at P =100 MPa. At that tantalum becomes more soluble in the melt than niobium. The pressure effect on columbite solubility within the range 30-400 MPa is less expressed. However at pressure decrease from 400 and 100 to 30 MPa resulted in a decrease in the maximal contents of Ta and Nb in the subaluminous melt by a factor of 3-6 (Chevychelov et al., 2010).
At the study conditions the diffusion coefficients of Ta, Nb and Mn in the melt are ~n·10-8 n·10-11 cm2/sec. The diffusion coefficients of Ta are minimal, the same of Nb are usually higher on half an order of magnitude, and the same of Mn are still higher (fig. 2). It is noted more appreciable influence of change of pressure and especially change of melt composition on values of diffusion coefficients in comparison with effect of temperature.
It is shown that the partition coefficients of Ta, Nb and Mn between the fluid and the granitic melt (weight ratio fluidCi/meltCi = fluid/meltDi) under the study conditions have very low values (0.001-0.008 for Ta, 0.001-0.022 for Nb, and 0.002-0.010 for Mn); that is these metals partition preferentially into the melt (fig. 3). The coefficients fluid/meltDTa and fluid/meltDNb generally increase either with increasing alumina ratio A/NKM in the glass composition, or with rising temperature. At our experimental conditions fluorine mainly concentrates in the melt: the partition coefficients of F are below 1, being within the range of 0.1-0.7. Peraluminous glasses have the maximum F content (up to ~4.5 -6 wt %) and in alkaline glasses the F content decreases up to ~0.5 -2 wt % (Borodulin et al., 2009).
Thus tantalum and niobium owing to their high melt affinity can not pass into a aqueous fluid in appreciable amounts and at fluid escape they are kept in the magmatic melt. It is experimentally shown that columbite solubility in subaluminous and peraluminous granitic melts has strongly expressed positive temperature dependence, that is with decrease of temperature the maximum Nb and especially Ta contents in these melts sharply decrease and in natural Li-F granitic melt with A/NK ~1.3 much stronger than in our synthetical melts. But nevertheless the columbite solubility in granitic melts remains enough high, especially in alkaline magmas. In terms of Ta and Nb concentrations, it is much higher than the abundances of these elements even in the most enriched granites. It can be safely concluded that the crystallization of columbite on the liquidus of both alkaline and alumina-rich magmas is impossible.
Tantalum and niobium apparently separate from the melt only at the end of melt crystallization at solidus temperature (550-600oC) with formation of the smallest dispersed accessory tantalite-columbites in intergrain space of the latest final differentiates of Li-F granites. However the content of magmatogene tantalum in Li-F granites and ongonites of the Kukulbei complex, Eastern Transbaikalia, and others rare-metal regions is in several times less than economic. Usually it does not exceed 30-50 ppm Ta2O5, at accepted boundary values 90 ppm in ore bodies of Orlovka and Etyka deposits and at the average contents 130-140 ppm in ores of these deposits. Apparently the further concentration of Ta up to an economic 100-350 ppm level occurs by the hydrothermal-metasomatic processes at greysening and albitization of Li-F granites with the help of postmagmatic fluids with redeposition of previous crystallized accessory tantalo-niobates in the upper parts of granite cupola beneath the screen of a hornfelsed schist roof (Chevychelov et al., 2005; Zaraisky et al., 2008).
This study was financially supported by RFBR grant No 08-05-00865-a.
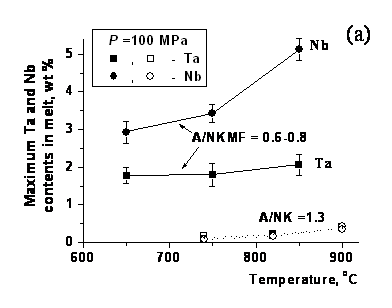

Fig. 1. Maximum Ta and Nb contents in glass at the boundary with columbite as a function of temperature (filled symbols). The aluminium saturation index, A/NKMF - Al2O3/(Na2O+K2O+MnO+FeO), in the melt ranges from (a) 0.6-0.8 to (c) 2.0-2.2. The lines show trends of variations in the maximum Ta and Nb contents in melt with temperature. For the sake of comparison, also shown are our previous results for the melt of natural Li-F granite with A/NK ~1.3 (unfilled symbols and dashed lines).

Fig. 2. The diffusion coefficients of Ta, Nb and Mn in the melt as a function of the melt composition.
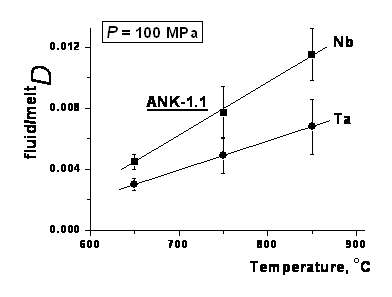
Fig. 3. The partition coefficients of Ta and Nb between the fluid and the granitic melt as a function of temperature.
References
Zaraisky G.P. Formation conditions of rare-metal deposits related to granitoid magmatism / Smirnov volume – 2004. Moscow: Smirnov Foundation, 2004. P. 105-192 (in Russian).
Chevychelov V.Yu., Zaraisky G.P., Borisovsky S.E., Borkov D.A. Effect of melt composition and temperature on the partitioning of Ta, Nb, Mn and F between granitic (alkaline) melt and fluorine-bearing aqueous fluid: fractionation of Ta and Nb and conditions of ore formation in rare-metal granites // Petrologiya, 2005. Vol. 13. № 4. P. 339-357 [Petrology, 2005. V. 13. No 4. P. 305-321].
Zaraisky G.P., Chevychelov V.Yu., Aksyuk A.M., Korzhinskaya V.S., Kotova N.P., Redkin A.F., Borodulin G.P.
Experimental substantiation of physical and chemical model of formation of tantalum deposits related to lithium-fluoric granites / Experimental researches endogenous processes. Memories of academician V.A. Zharikov. Chernogolovka: Publishing department of IPCP RAS, 2008. P. 86-109 (in Russian).
Borodulin G.P., Chevychelov V.Yu., Zaraisky G.P. Experimental study of partitioning of tantalum, niobium, manganese, and fluorine between aqueous fluoride fluid and granitic and alkaline melts // Doklady Akademii Nauk. 2009. Vol. 427. № 2. P. 233-238 [Doklady Earth Sciences. 2009. Vol. 427. No 5. P. 868-873].
Chevychelov V.Yu., Borodulin G.P., Zaraisky G.P. Solubility of columbite, (Mn,Fe)(Nb,Ta)2O6, in granitoid and alkaline melts at 650-850oC and 30-400 MPa: an experimental investigation // Geokhimiya. 2010. № 5. P. 485-495 [Geochemistry International. 2010. Vol. 48. No 5. P. 456-464].