2010
| News | Registration | Abstracts | Accommodation | Excursions | Deadlines | Organizing committee |
| First circular | Participants | Abstract submission | Travel | Program | Seminar History | Contact us |
| Новости |
| Первый циркуляр |
| Регистрация |
| Оформление тезисов |
| Тезисы |
| Программа |
| Участники |
| Размещение |
| Экскурсии |
| Проезд |
| Важные даты |
| Оргкомитет |
| Обратная связь |
Ce+4/ Ce+3 variations in magmatic and secondary zircons from alkaline rocks as a reflection of differences in the oxygen fugacity
Yu.A. Balashov, E.V. Martynov, L.G. Balashova
Geological Institute, KSC RAS, Apatity, Russia
Significant variations of the oxygen fugacity in the mantle rocks affect redistribution of the elements with variable valence (Fe, Eu, V, Cr…) in rocks and minerals. It also concerns accessory zircons. As already noted (Balashov, Skublov, 2009), two valence forms - Ce+4 and Ce+3 are present as an isomorphous admixture in zircon structures. This allowed one to propose that their ratio is controlled by the real level of the oxygen fugacity at the zircon formation which should agree with the P-T-ƒO2 petrological parameters of the initial melt (or solution). A correlation has been revealed in comparing the calculated Ce+4/ Ce+3 in zircon from numerous kimberlitic pipes, lamproites and deep-seated mantle xenolith with the data on the ilmenite buffer FMQ of the kimberlitic pipes (Ashchepkov et al, 2008) and on the spinel buffer FMQ (Ballhaus, 1993). The result is presented in Fig. 1. A relationship between Ce+4/ Ce+3 and buffer FMQ is described by the linear model y=14.742+8.954*x (x is the buffer, y is the ratio Ce+4/ Ce+3 in zircon). Explicit data, as a first approximation, on Ce+4/Ce+3 in zircon and on rock ∆logƒO2 for the upper mantle correlate well and embrace a considerable variation range from 27 to 0.01 in zircon and from +1.7 to – 2 in ilmenite (Fig. 1).
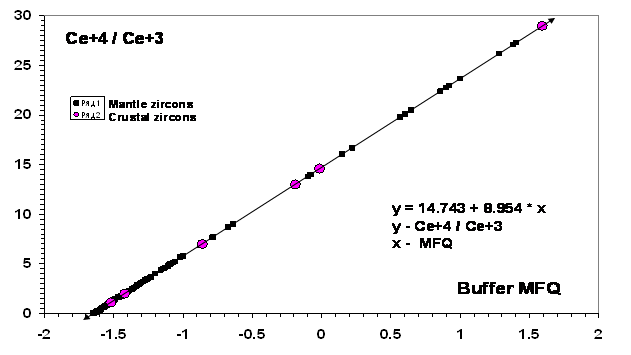
Fig. 1. The proposed correlation between the oxygen fugacity variations FMQ = ∆logƒO2 in mantle and crustal rocks and the changes in the Ce+4/ Ce+3 ratio in zircons from these rocks.
Since the ratio Ce+4/Ce+3 is defined with a much higher precision (± 0.1 – 0.01) than for the real gradation by the buffer FMQ (≈ ± 1-0.1), the use of a new buffer Ce+4/Ce+3 seems to be rather promising. According to Fig. 1, the supposed zero value of the buffer FMQ agrees with Ce+4/Ce+3 ≈ 14.7 in zircons. A shift in the direction of the Ce+4 excess is caused by the closeness of the ionic radius Ce+4(0.97Å) to Zr+4(0.84Å) as compared with Ce+3 (1.143Å). A study of the data on the Ce+4/ Ce+3 variations in the zircons from kimberlite showed that xenolith of the Chomur (Yakutia) pipe has uniquely high ratios (23 -16) that are not found in other kimberlitic pipes (7.7 – 0.02). Such high for cerium ratios (27.3- 13.8) are noted in lamproites of the Argyle (Australia) pipe. This agrees with Ce+4/Ce+ 3 for the zircons from crustal granitoids and Hadean detrital zircons from Australia (29 – 13) (Balashov, Skublov, 2009). Such a coincidence points to the contamination probability or participation of the crustal material in one form or another. Besides, it is worth noting that the ∆logƒO2 values change in the direction of the positive values > 0.5-1.0 in the upper parts of the kimberlitic pipes confined to the transition zone between garnet and spinel peridotite of the lithosphere ( P ≈ 30-20 kbar ) (Ashchepkov et al, 2008 ) .
As a result, the correlation range is substantially extended and, moreover, it combines the variation regularities of the oxygen fugacity in the upper mantle and crust. Hence it follows that the participation of the crustal material is an “acidification” factor for the upper lithosphere. At the same time the data on the Ce secondary zircon from granitoids have very low Ce+4/Ce+3 (Balashov, Skublov, 2009) which corresponds to the majority of the lithospheric peridotite xenoliths (Ashchepkov et al, 2008).
Analysing fugacity of other alkaline rocks we should emphasize that the revealed Ce+4/Ce+3 ratios appeared to be very contrasting (Fig. 2). Larvikite of Norway and carbonatite of Kovdor are characterized by a reduction regime that is comparable with mantle deep layers of the lithosphere. On the contrary, extremely high values of the oxidizing regime are noted in syenite and Ne-syenite pegmatites of Norway. They exceed the mantle level by 1-3 orders (Fig. 3). If this effect is caused by exposure to the atmosphere in one form or another, then it points to the significance of the study of fugacity processes in the crust. In particular, this is of special interest for revealing secondary geochemical processes of contamination in magmatic systems of the lithosphere upper zones and for an assessment of the evolution of the ore formation conditions in the crust.
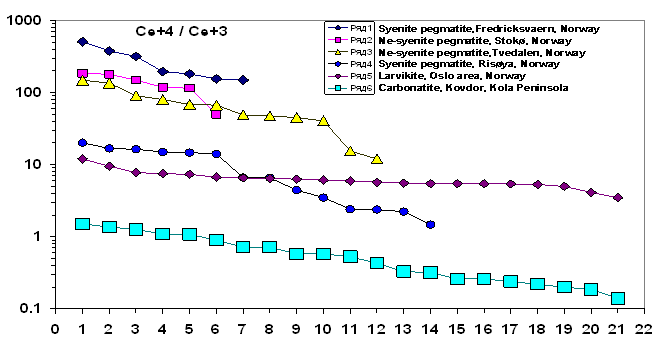
Fig.2. Ce+4/Ce+3 variations in zircons from alkaline rocks and pegmatite.
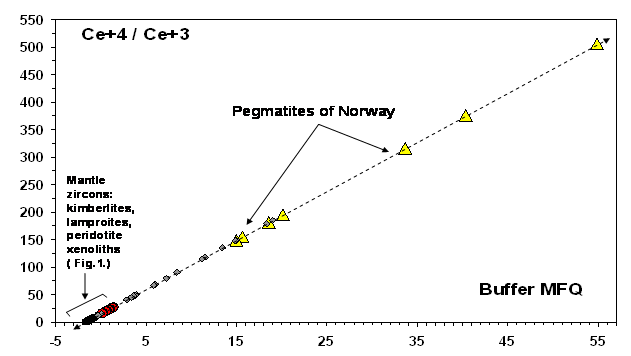
Fig. 3. A general range of Ce+4/Ce+3 variations in mantle and crust zircons including the data on alkaline rocks (Belousova E.A. et al, 1998, 2002). The data for the mantle are divided into two groups – with reduced and increased values by the buffer MFQ.
The authors are grateful to Belousova E.A. for the granted possibility to use a part of the collection of REE from zircons.
References:
Balashov Yu. A., Skublov S.G. Uncommon indicatory possibility cerium in zircons of different genesis // “Physicochemical factors of petrogenesis and formation of ore deposits”, dedicated to the 110th anniversary of the great Russian petrologist Dmitry Sergeevich Korzhinsky. M. IGEM RAS, 2009. P. 67-70.
Ashchepkov I.V., Pokhilenko N.P., Vladykin N.V., Logvinova A.M., Rotman A.Y., Afanasiev V.P., Pokhilenro L.N., Kuligin S.S., Malygina L.V., Alymova N.V., Stegnitsky Y.B., Khmelnikova O.S. Plum interaction and evolution of continental mantle lithosphere // Deep-seated magmatism, its sources and plumes: Proceedings of VIII International Workshop. Vladivostok-Irkutsk. 2008. P. 104-121.
Ballhaus C. Redox states of lithospheric and asthenospheric upper mantle // Contrib.Mineral.Petrol. 1993. V.114. P. 331-348.
Belousova E.A., Griffin W.L., O’Reilly S. Y., Fisher N.I. Igneous zircon: trace element composition as an indicator of source rock type // Contrib. Mineral. Petrol. 2002. V.143. P. 602-622.
Belousova E.A., Griffin W.L., Pearson N.J. Trace element composition and cathodoluminescece properties of southern Africa kimberlitic zircons // Mineral. Magazine. 1998. V. 62. P. 355-366.